This equation states that the emf is equal to the change in free energy per unit charge (nF). The emf of the cell, E, can be related to the change in free energy by the equation: The negative sign indicates that the reaction is spontaneous, and the magnitude of ΔG° is related to the amount of work that can be obtained from the reaction. Where ΔG° is the change in Gibbs free energy, n is the number of electrons transferred, F is the Faraday constant (the charge on one mole of electrons), and E° is the standard electrode potential. The change in free energy for this reaction can be expressed as: The electrons flow from the zinc electrode (anode) to the copper electrode (cathode) through an external circuit. This reaction involves the transfer of electrons from zinc to copper ions, which results in the formation of zinc ions and copper metal. The cell emf, or electromotive force, can be derived using the principles of thermodynamics and electrochemistry.įirst, consider a redox reaction that occurs spontaneously at standard conditions, such as the reaction between zinc and copper ions: Once the emf of a cell is known, it can be used to calculate the current flowing through a circuit connected to the cell, using Ohm's law and Kirchhoff's laws. These values can be found in standard electrode potential tables. Where E(copper) is the reduction potential of the copper electrode and E(zinc) is the oxidation potential of the zinc electrode. These potentials are measured relative to a standard hydrogen electrode (SHE) under standard conditions of temperature, pressure, and concentration.įor example, if a cell has a zinc electrode (anode) and a copper electrode (cathode), the emf of the cell can be calculated as follows: The reduction potential and oxidation potential are standard electrode potentials that represent the tendency of an electrode to gain or lose electrons, respectively. Where E(cathode) is the reduction potential of the cathode and E(anode) is the oxidation potential of the anode. The cell emf can be calculated using the following formula: 
The emf of a cell can also be affected by external factors such as the resistance of the circuit connected to the cell, which can lead to a decrease in the current flowing through the circuit. The emf of a cell depends on various factors, such as the type of electrodes and electrolyte used in the cell, temperature, and concentration of the electrolyte. 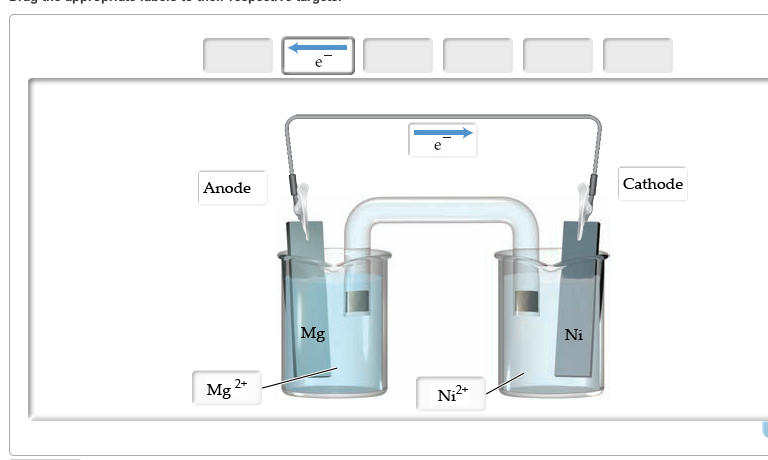
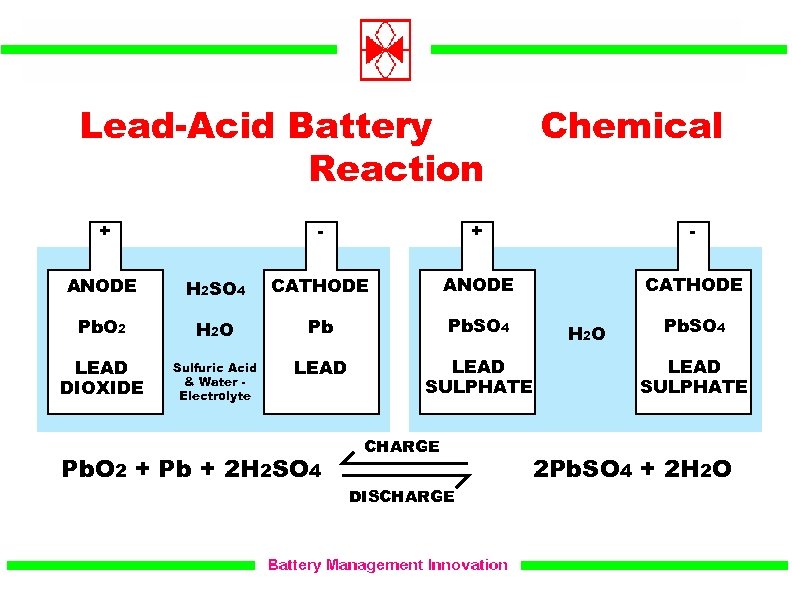
It is a measure of the difference in electrical potential between the two terminals of the cell, and is typically expressed in units of volts (V). In other words, cell emf is the driving force behind the flow of electric current in a circuit. It is essentially the voltage produced by a cell, which drives the flow of electrons from the negative terminal (anode) to the positive terminal (cathode) of the cell. Percentage Concentration To Molarity CalculatorĬell emf, or electromotive force, is a measure of the energy per unit charge that is supplied by a cell or battery to push electrons through an external circuit.To determine oxidation electrodes, the reduction equation can simply be flipped and its potential changed from positive to negative (and vice versa). The table below is a list of important standard electrode potentials in the reduction state. Important Standard Electrode (Reduction) Potentials We could have accomplished the same thing by taking the difference of the reduction potentials, where the absent or doubled negation accounts for the fact that the reverse of the reduction reaction is what actually occurs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
